Twisted or shifted? Fluorescence measurements of late intermediates in transcription initiation by T7 RNA polymerase
Rosemary S. Turingan, Karsten Theis, & Craig T. Martin, Biochemistry 46, 6165-6168, 2007.
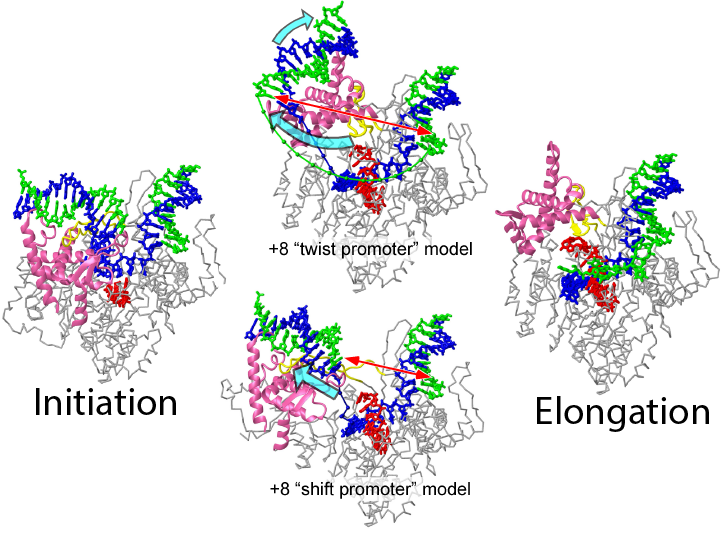
T7 RNA polymerase undergoes dramatic structural rearrangements in the transition from initiation to elongation. Two models have been proposed for promoter-bound intermediates late in the transition. (i) A subset of promoter interactions are maintained through completion of the protein conformational (twist) change, and (ii) concerted movement (shift) of all promoter-binding elements away from the growing DNA-RNA hybrid leads to an open intermediate, with large-scale domain rotations deferred until after promoter release. Fluorescence resonance energy transfer measurements provide very strong support for the latter.
PMID: 17472344 PMCID: PMC2546521 DOI: 10.1021/bi700058b